Imagine developing a generic drug without ever giving it to a single human volunteer. No blood draws. No hospital stays. No expensive clinical trials. Just a machine that dissolves a pill in a lab, and a computer that says: “This will behave exactly like the brand-name drug in your body.” That’s not science fiction. It’s IVIVC - and it’s changing how generic drugs get approved.
For decades, proving that a generic drug works the same as the original meant running bioequivalence studies. You’d give 24 to 36 healthy volunteers the brand drug, then give them the generic a few weeks later. You’d draw blood every 30 minutes for 24 hours. You’d measure how much drug entered the bloodstream. You’d compare the curves. It cost between $500,000 and $2 million per study. And it took months. For companies making dozens of generics, that added up fast.
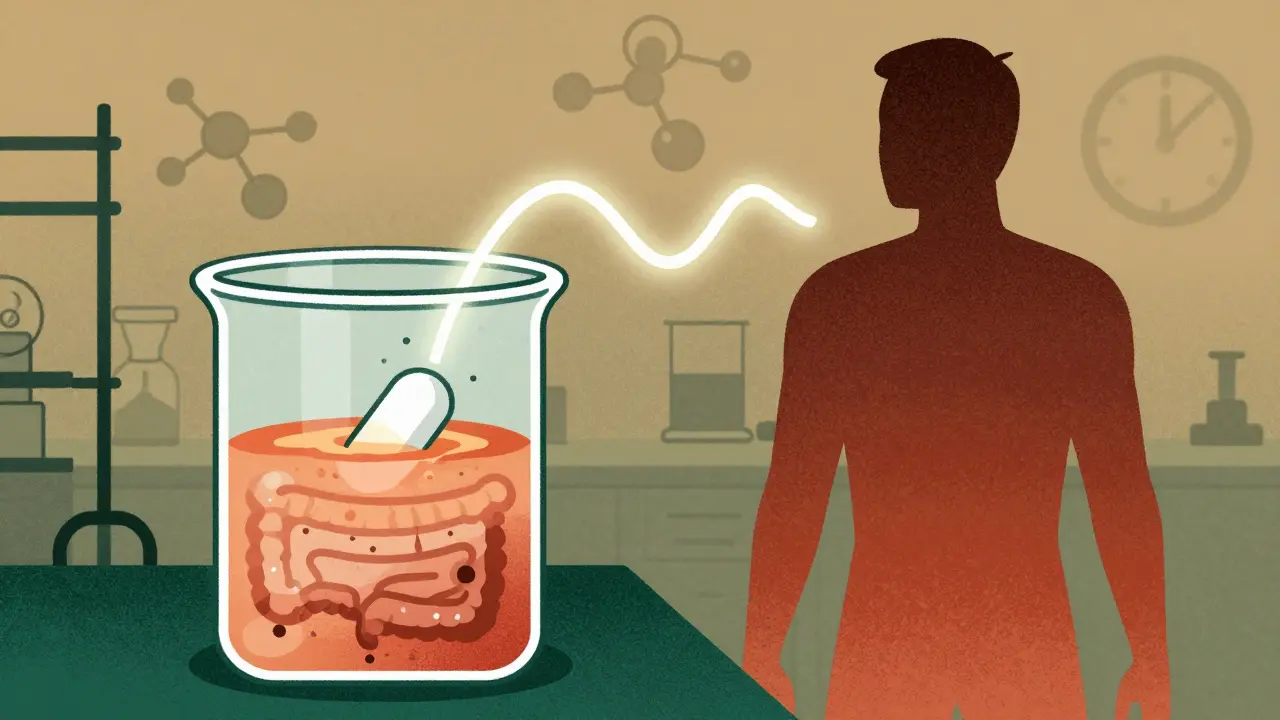
Then came IVIVC - In Vitro-In Vivo Correlation. It’s not just a fancy acronym. It’s a scientific bridge. It links what happens to a pill in a test tube (in vitro) to what happens in your bloodstream (in vivo). If you can prove that the way a drug dissolves in a lab accurately predicts how it’s absorbed in your gut, then you don’t need to test it on people. You get a biowaiver. And that’s a huge deal.
How IVIVC Works: From Dissolution to Bloodstream
At its core, IVIVC is about prediction. You start with dissolution testing. You put the drug in a vessel that mimics the stomach and intestines - not just water, but fluid with the right pH, bile salts, and enzymes. You stir it. You measure how much drug comes out over time. That’s your in vitro profile.
Then you run real human studies - but not just one. You test multiple versions of the drug. A fast-release version. A slow-release version. Maybe even one with a slightly different coating. You collect blood samples from each volunteer at 12 or more time points. You plot the concentration curves. You compare those curves to the dissolution profiles.
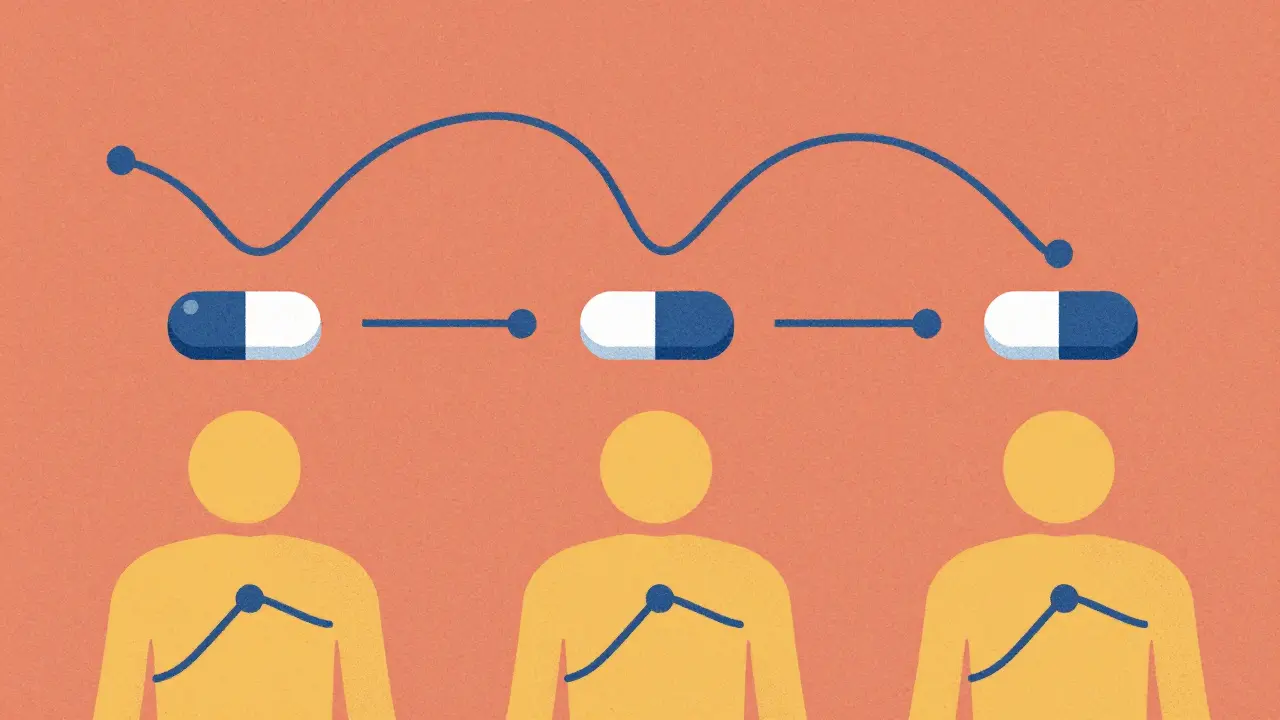
The goal? Find a mathematical match. If the shape of the dissolution curve mirrors the shape of the blood concentration curve - point by point - you’ve got what’s called a Level A correlation. That’s the gold standard. It means you can take a dissolution result from a new batch of pills and say with confidence: “This will produce the same AUC and Cmax as the reference product.” AUC is total exposure. Cmax is the peak concentration. Both matter for safety and effectiveness.
The FDA says a valid Level A model must have an R² value over 0.95. That’s 95% of the variation in blood levels explained by the dissolution data. The model must predict within ±10% for AUC and ±15% for Cmax. That’s tight. But when it works, it’s powerful.
Why IVIVC Matters: Saving Time, Money, and Lives
The numbers speak for themselves. Avoiding one bioequivalence study saves $1 to $2 million. That’s not a rounding error. That’s the difference between a generic drug being profitable or shelved. For companies making extended-release products - like opioids, blood pressure meds, or ADHD treatments - IVIVC isn’t optional. It’s essential.
Take Teva’s experience with their extended-release oxycodone generic. They spent 14 months and three formulation tries to build a working IVIVC. But once they got it, they avoided five full bioequivalence studies. That’s over $5 million saved. And faster approvals mean patients get affordable access sooner.
But IVIVC isn’t just about cost. It’s about ethics. Human testing isn’t dangerous, but it’s not risk-free. Blood draws. Fasting. Hospital visits. For drugs with narrow therapeutic windows - where too little doesn’t work and too much causes toxicity - minimizing human exposure is a win.
And it’s not just for new drugs. IVIVC lets manufacturers make small changes without restarting clinical trials. Need to switch suppliers for an excipient? Move production to a new facility? Adjust the tablet weight by 3%? Without IVIVC, each change requires a new study. With it? You run a dissolution test. If it matches, you’re approved.
The Four Levels of IVIVC - And Why Level A Is King
Not all correlations are created equal. The FDA classifies them into four levels:
- Level A: Point-to-point match. Dissolution at 10 minutes = blood concentration at 10 minutes. Dissolution at 30 minutes = blood concentration at 30 minutes. This is the only level that allows full biowaivers for all changes. It’s rare. But when you have it, you’re golden.
- Level B: Uses average values. You compare mean dissolution time to mean residence time. It’s less precise. You can’t predict individual curves. Regulatory agencies accept it for some cases, but not for major changes.
- Level C: One point only. Maybe dissolution at 1 hour correlates with Cmax. That’s it. You can’t predict AUC. It’s easier to build but risky. The EMA and FDA warn it might miss important differences in absorption.
- Multiple Level C: You link several dissolution time points to several pharmacokinetic parameters. Better than single Level C, but still not as reliable as Level A.
For biowaivers, regulators almost always want Level A. It’s the only one that gives you full confidence. Level C might get you through the door for a minor change, but if you’re planning long-term flexibility, you need Level A.
Why Most IVIVC Submissions Fail
Despite the benefits, only about 42% of IVIVC submissions get approved - up from 15% in 2018, but still low. Why? Three reasons dominate:
- Wrong dissolution conditions. If you test the drug in plain water at pH 7, you’re not simulating the gut. Real absorption happens in a complex environment with bile, enzymes, and varying pH. Biorelevant media - with bile salts and surfactants - are now standard. Over 64% of failed submissions in 2023 were rejected for using outdated, non-physiological methods.
- Too few formulations. You can’t build a model from one or two versions. You need at least three: fast, medium, and slow. Each must produce a different dissolution profile. If all your tablets dissolve the same way, the model has no data to correlate. 74% of failures came from insufficient formulation space.
- Poor validation. A model that fits your data doesn’t mean it works on new data. You must test it on a separate batch - one you didn’t use to build the model. If it fails that test? Back to square one. 68% of rejections were due to weak validation strategies.
And it’s not just technical. You need high-quality pharmacokinetic data. That means dense sampling - 12+ time points per subject - and enough subjects (12-24 per study) to capture variability. Most small companies don’t have the resources. That’s why contract labs like Alturas Analytics and Pion now handle 60-70% of successful IVIVC projects. They’ve got the expertise. The equipment. The experience.
When IVIVC Doesn’t Work - And What to Do Instead
IVIVC isn’t magic. It fails for some products:
- Narrow therapeutic index drugs - like warfarin, digoxin, or cyclosporine. Even tiny differences matter. Regulators won’t waive human testing.
- Non-linear pharmacokinetics - where doubling the dose doesn’t double the blood level. Too complex for a simple dissolution model.
- Drugs with food effects - if absorption changes drastically with meals, and your dissolution test doesn’t account for it, the model will be wrong.
- Topical, injectable, or ophthalmic products - until recently, IVIVC was only for oral drugs. But in 2023, the FDA released draft guidance for topical products. The EMA is exploring it for injectables. It’s coming - but it’s still early.
For immediate-release drugs, there’s a simpler path: the Biopharmaceutics Classification System (BCS). If your drug is Class I - highly soluble and highly permeable - you might qualify for a biowaiver without IVIVC. But for extended-release, modified-release, or complex formulations? IVIVC is your only option.

The Future of IVIVC: Machine Learning and Global Harmonization
The field is evolving fast. In 2024, the FDA and EMA held a joint workshop on machine learning for IVIVC. Instead of linear regression models, they’re now testing AI algorithms that can find patterns in dissolution and pharmacokinetic data no human would spot. Early results show promise - especially for products with multiple release mechanisms.
Also, biorelevant dissolution is becoming standard. By 2025, 75% of new IVIVC submissions are expected to use media that mimic the human GI tract. That’s a big shift from the old USP water-and-paddle method.
And the numbers are climbing. The global dissolution testing market hit $487 million in 2022 and is growing at 6.2% a year. Why? Because more companies are realizing: if you don’t invest in IVIVC, you’ll be stuck paying for endless bioequivalence studies. The winners are those who build the model early - during Phase 2 trials for new drugs, or during prototype development for generics.
By 2027, McKinsey predicts IVIVC-supported waivers will cover 35-40% of all modified-release generic approvals - up from 22% today. That’s not just efficiency. It’s a revolution in how we prove drugs work.
Final Takeaway: IVIVC Is Hard - But Worth It
Building a valid IVIVC isn’t easy. It takes time. Money. Expertise. And a willingness to fail - often - before you get it right. But for companies serious about making complex generics, it’s the smartest investment they can make.
It’s not about cutting corners. It’s about using science to replace unnecessary human testing. It’s about faster access to affordable medicines. It’s about smarter regulation.
The future of bioequivalence isn’t more volunteers. It’s better models. Better data. Better science. And IVIVC is leading the way.
What is IVIVC and how does it relate to biowaivers?
IVIVC stands for In Vitro-In Vivo Correlation. It’s a scientific model that links how a drug dissolves in a lab (in vitro) to how it’s absorbed in the human body (in vivo). If the model is strong enough - usually a Level A correlation - regulators like the FDA and EMA can approve a biowaiver. This means the drug doesn’t need human bioequivalence studies. Instead, dissolution testing alone proves the generic behaves like the brand-name product.
Why is Level A IVIVC the only one accepted for full biowaivers?
Level A IVIVC provides a point-to-point match between dissolution and blood concentration over time. That means you can predict the entire pharmacokinetic profile - including AUC and Cmax - from dissolution data alone. Other levels (B, C) only give partial predictions. For regulatory agencies, that’s not enough confidence. A full biowaiver requires certainty that every batch will perform the same in patients. Only Level A delivers that.
What are the biggest reasons IVIVC submissions get rejected?
The top three reasons are: 1) Using non-biorelevant dissolution media (like plain water instead of simulated gut fluid), 2) Not testing enough different formulations (you need at least three with distinct release rates), and 3) Failing to properly validate the model on independent data. Over 64% of 2023 rejections were due to poor physiological relevance of the dissolution method.
Can IVIVC be used for all types of drugs?
No. IVIVC works best for oral, modified-release products with predictable absorption. It’s not accepted for drugs with narrow therapeutic windows (like warfarin), non-linear pharmacokinetics, or strong food effects. Topical, injectable, and ophthalmic products are still under evaluation. For immediate-release drugs, the BCS classification often provides a simpler biowaiver route.
How much does it cost to develop an IVIVC model?
Developing a Level A IVIVC typically costs between $800,000 and $1.5 million over 12-18 months. That includes formulation development, multiple pharmacokinetic studies, dissolution method optimization, and modeling. But it saves $1-2 million per avoided bioequivalence study. For companies with multiple products, the return on investment is clear.
Is IVIVC used globally, or just in the U.S.?
IVIVC is accepted by both the U.S. FDA and the European Medicines Agency (EMA), and increasingly by regulators in Canada, Japan, and Australia. The EMA’s 2021 reflection paper shows growing interest, with 23% of scientific advice requests now involving IVIVC. While the requirements vary slightly, the core principles are harmonized. Global adoption is rising, especially for complex generics.





9 comments
Lebogang kekana
Let me tell you something - this IVIVC stuff is a game-changer. I work in pharma logistics in Johannesburg, and I’ve seen how long it takes to get generics approved the old way. Months. Millions. Deadlines slipping. Now? One dissolution test and boom - approval. I’ve watched companies go from bankrupt to profitable because of this. No more human trials for every little tweak. It’s not just science - it’s survival.
And don’t get me started on how many patients in developing countries wait for life-saving meds. IVIVC doesn’t just save money. It saves lives. Real ones. Not theoretical. Real.
Jessica Chaloux
OMG I’m crying 😭 this is literally the most beautiful thing I’ve read all year. Like… imagine not having to stick needles in people just to prove a pill works? That’s peace. That’s love. That’s science with a soul. 🌈💖
Mariah Carle
There’s a deeper metaphysical layer here, isn’t there? IVIVC isn’t just a technical model - it’s a rejection of the Cartesian dualism between body and machine. The pill dissolves in a beaker, yet it ‘knows’ how to behave in the human gut. That’s not correlation - that’s resonance. We’re witnessing the dissolution of the sacred boundary between laboratory and life. The body, reduced to data. The machine, elevated to oracle.
And yet… who are we to say what’s ‘natural’? If a pill’s journey through a simulated intestine predicts its path through a real one - is that not more honest than injecting strangers with drugs for statistical comfort? We’ve replaced spectacle with symmetry.
Philosophy doesn’t stop at the lab door. It walks right in - and brings a pipette.
Justin Rodriguez
Just want to clarify one thing I see people miss: Level A isn’t just about R² > 0.95. It’s about reproducibility across labs. I’ve seen companies build models that work perfectly in-house - then fail when the FDA runs their own dissolution tests. The key is using biorelevant media consistently. Bile salts aren’t optional. pH gradients aren’t suggestions. And you need at least three formulations - fast, medium, slow - or your model has zero predictive power.
Also - don’t skip validation. Test on a batch you didn’t train on. If it fails? You didn’t build a model. You memorized noise.
It’s not magic. It’s rigor. And it’s worth every hour.
Raman Kapri
You all sound like you’re celebrating a magic pill. But let’s be honest - this is just corporate efficiency dressed up as ethics. Where’s the transparency? Who verifies these models? Who ensures the dissolution machines are calibrated? Who checks if the ‘biorelevant media’ is just a fancy version of tap water with food coloring?
And don’t pretend this isn’t about cutting corners. $5 million saved? That’s not saving lives - that’s increasing shareholder value. The patients still get the same drug. The regulators still approve it. But now, the process is hidden behind proprietary algorithms and contract labs.
Don’t confuse cost-cutting with progress. This isn’t science. It’s accounting with a lab coat.
Megan Nayak
Oh wow. So now we’re trusting computers to predict human biology? How quaint. Let me guess - next they’ll replace doctors with chatbots and nurses with AI-powered IV drips. This ‘IVIVC’ is just the first step toward pharmaceutical eugenics. If you can’t test on humans, how do you know what happens in people with liver disease? Or pregnancy? Or diabetes? Or depression? Or gut dysbiosis?
Every human body is a unique ecosystem. You can’t reduce that to a dissolution curve. This isn’t innovation. It’s arrogance. And it’s going to kill someone. Mark my words.
Also - why are we still using AUC and Cmax? Those are 1970s metrics. We need metabolomics. We need microbiome data. We need epigenetics. But no - let’s just plug numbers into a regression and call it a day. Pathetic.
Tildi Fletes
While the theoretical framework of IVIVC is robust, the practical implementation requires extraordinary precision. The most critical oversight in submissions remains the lack of statistical power in pharmacokinetic sampling. Many studies utilize fewer than 12 subjects, with sparse time-point sampling (e.g., 6–8 time points), which fundamentally undermines the ability to model absorption kinetics with sufficient fidelity.
Additionally, the assumption of linearity in dissolution behavior across all pH environments remains empirically unsupported for many modern formulations. The use of biorelevant media must be accompanied by dynamic pH profiling - not static buffers - to reflect the physiological gradient from stomach to ileum.
Validation must include cross-laboratory reproducibility testing. Without it, the model is not generalizable. And without generalizability, biowaivers are not scientifically defensible.
These are not minor details. They are foundational.
Betsy Silverman
I work in rural pharmacy in Nebraska. We get generics from everywhere. Some work. Some don’t. I’ve had patients come in saying their new pill ‘doesn’t feel right.’ No lab test could catch that - but maybe IVIVC could, if it’s done right.
I’m not against this. I’m just scared it’ll be used to cut corners instead of lifting standards. If we’re going to replace human trials, we need to replace them with something better - not just cheaper.
Can we make sure the dissolution machines are checked daily? Can we make sure the labs aren’t just using the cheapest media? Can we audit this like we audit blood banks?
Because if not - people will die quietly. And no one will know why.
Zacharia Reda
So… you spent $1.2 million to avoid paying $5 million in clinical trials? That’s not innovation. That’s a financial hedge. And you’re calling it ‘science’?
Meanwhile, the guy in India who can’t afford his blood pressure med still can’t get it because the generic manufacturer didn’t bother with IVIVC - they just slapped a label on a pill and shipped it. So who’s really benefiting here?
Also - why is no one talking about the fact that 70% of successful IVIVC submissions come from just 3 contract labs? That’s not democratizing science. That’s creating a cartel.
Brilliant engineering. Terrible ethics. Classic.