Why Your Generic Pill Might Look Different
You walk into the pharmacy, hand over your prescription, and the pharmacist hands you a bottle. The label says the same drug name, but the pill inside looks completely different. It's a different color, maybe a different shape, and definitely not the one your doctor showed you in the brochure. This is a common moment of doubt for many patients. You wonder if this new, cheaper version will actually work the same way as the brand-name version you were used to. The answer lies in a specific scientific term you might see on the label or in medical discussions: bioequivalence.
Understanding bioequivalent medications is drugs that perform similarly in the human body to a reference product, ensuring the same therapeutic effect isn't just about saving money. It's about safety and trust. When you switch from a brand-name drug to a generic, you are relying on a rigorous regulatory process designed to prove that the new version delivers the same amount of medicine to your bloodstream, at the same speed. This concept is the backbone of the modern pharmaceutical market, allowing access to life-saving treatments at a fraction of the original cost without compromising health outcomes.
The Science Behind the Label
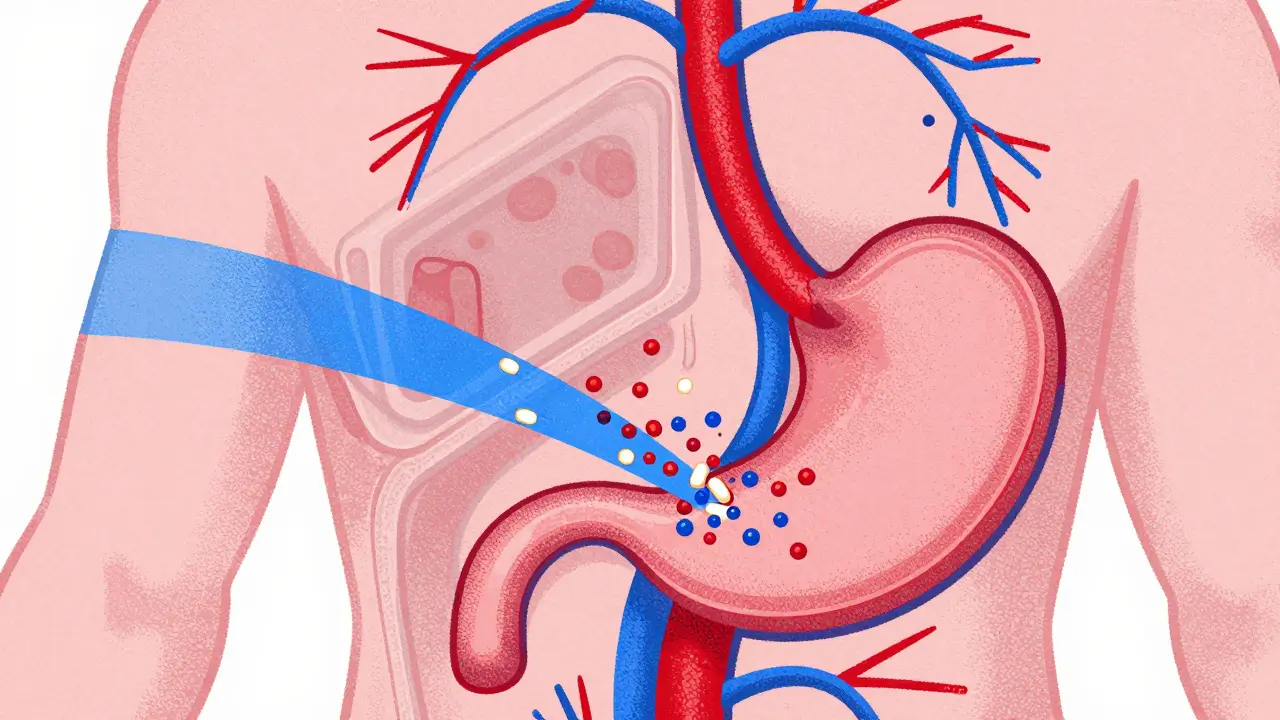
To understand bioequivalence, you first need to understand how your body handles a drug. When you swallow a tablet, it doesn't instantly heal you. It has to dissolve, pass through your digestive system, and enter your bloodstream. This process is called pharmacokinetics. Regulatory agencies like the FDA is the U.S. Food and Drug Administration, the federal agency responsible for protecting public health by regulating drugs and medical devices measure exactly how much of the active ingredient gets into your blood and how fast it gets there.
Two drugs are considered bioequivalent if they show no significant difference in the rate and extent to which the active ingredient becomes available at the site of drug action. In plain English, this means the generic drug and the brand-name drug act the same way in your body. They don't need to look the same or contain the same inactive ingredients (like fillers or dyes). They just need to deliver the active medicine effectively.
This standard wasn't always in place. The modern framework for approving generic drugs was established by the Hatch-Waxman Act is the Drug Price Competition and Patent Term Restoration Act of 1984, which created the pathway for generic drug approval in the United States. This legislation balanced the need for innovation with the need for affordable access. It allowed generic manufacturers to skip the expensive, lengthy clinical trials required for brand-new drugs, provided they could prove their product was bioequivalent to the already-approved brand-name version.
How Bioequivalence is Measured
So, how do regulators actually prove two drugs are the same? They don't just guess. They conduct specific studies involving healthy volunteers. These studies track the concentration of the drug in the blood over time. There are three key numbers that scientists look at:
- AUC (Area Under the Curve): This represents the total amount of drug exposure your body gets over time. It tells us how much of the medicine actually got absorbed.
- Cmax (Maximum Concentration): This is the peak level of the drug in your blood. It matters because if the peak is too high, you might get side effects. If it's too low, the drug might not work.
- tmax (Time to Maximum Concentration): This is how long it takes to reach that peak level. For some drugs, speed matters (like pain relievers), while for others, a steady release is better.
For two products to be approved as bioequivalent, the FDA requires that the 90% confidence interval of the generic drug's AUC and Cmax compared to the brand-name drug falls within a specific range: 80% to 125%. This might sound like a wide gap, but it is statistically derived. The assumption is that a difference of up to 20% in drug exposure is not clinically significant for most medications. In reality, most generic drugs fall much closer to the brand-name standard, often within 90% to 110% of the reference product.
| Parameter | What It Measures | Why It Matters |
|---|---|---|
| AUC | Total drug exposure over time | Ensures the total dose absorbed is sufficient for treatment |
| Cmax | Peak blood concentration | Prevents toxicity from sudden high spikes or failure from low peaks |
| tmax | Time to reach peak concentration | Important for drugs where speed of onset is critical |
Bioequivalence vs. Therapeutic Equivalence
It is easy to mix up terms, but there is a distinct difference between bioequivalence and therapeutic equivalence. Bioequivalence is a measure of how the drug moves through your body. Therapeutic equivalence is the conclusion that the drugs will produce the same clinical result. The FDA assigns codes to drugs in their Orange Book is the official publication listing all approved drug products with therapeutic equivalence evaluations to help pharmacists know which generics can be substituted.
A drug rated "AB" in the Orange Book is therapeutically equivalent. This means it is both pharmaceutically equivalent (same active ingredient, strength, and dosage form) and bioequivalent. This is the gold standard for substitution. If a generic has an "A" rating, you can expect it to work just as well as the brand-name drug. However, not all generics get this rating. Some might be bioequivalent but differ in other ways, such as the route of administration or specific formulation requirements that affect safety.
Pharmaceutical equivalence is the baseline. It means two products contain the same active ingredient in the same strength and dosage form. But two pills can be pharmaceutically equivalent without being bioequivalent. Imagine two cars with the same engine and tires. If one has a clogged fuel line, it won't run the same way even if the parts look identical. Bioequivalence testing ensures the "fuel line" isn't clogged and the engine runs smoothly.
When Bioequivalence Matters Most
For the vast majority of medications, the 80-125% standard works perfectly. Generic drugs account for approximately 90% of all prescriptions dispensed in the United States, and they are the backbone of affordable healthcare. However, there are exceptions. Some drugs have a Narrow Therapeutic Index is a category of drugs where the difference between a therapeutic dose and a toxic dose is very small (NTI). These are medications where a small change in blood concentration can lead to treatment failure or toxicity.
Examples of NTI drugs include Levothyroxine is a thyroid hormone replacement medication often used as a case study for bioequivalence concerns, certain anti-seizure medications, and some heart medications. For these drugs, the FDA sometimes requires tighter equivalence limits, such as 90% to 111%, instead of the standard 80% to 125%. This is because a 20% difference could be dangerous for a patient relying on precise hormone levels or seizure control.
Patients taking these specific medications often report feeling different when they switch manufacturers. While the FDA maintains that current standards are sufficient, some experts and patient groups advocate for stricter consistency in these specific categories. If you are on an NTI drug, it is often recommended to stick with the same manufacturer once you find a version that works well for you. Pharmacists can usually help with this by checking the "dispense as written" status on your prescription.
Cost, Savings, and Market Impact
The reason we have bioequivalence standards is largely economic. Developing a new brand-name drug costs billions of dollars and takes over a decade. Generic manufacturers don't need to repeat the safety and efficacy trials, only the bioequivalence studies. This significantly reduces the cost of bringing a drug to market. The average cost to develop a generic drug, including bioequivalence studies, is approximately $2.2 million. Compare that to the billions spent on brand-name development, and the price difference becomes clear.
Generic drugs save the U.S. healthcare system an estimated $2.2 trillion over the past decade. For an individual patient, this translates to savings of approximately $313 per prescription on average. This accessibility is vital. Without the bioequivalence pathway established by the Hatch-Waxman Act, many patients would simply not be able to afford their chronic medications. The global generic pharmaceuticals market was valued at over $400 billion in 2022, showing the massive scale of this industry.
However, the market isn't perfect. Sometimes, a generic manufacturer changes their production site or formulation slightly, which can lead to minor variations in how the drug performs. While these changes still meet regulatory standards, sensitive patients might notice them. This is why the FDA monitors adverse event reports closely. Data from the FDA's Adverse Event Reporting System shows that reports of problems with generic drugs are proportional to their market share, suggesting that they are generally as safe as brand-name options.
Common Myths and Real Experiences
There is a persistent myth that generics are "lower quality" or "watered down" versions of brand drugs. This is incorrect. Generic drugs must meet the same rigorous quality standards as brand-name drugs. They are manufactured in facilities inspected by the FDA to ensure consistency and purity. The only differences allowed are in inactive ingredients, which might affect the color, shape, or taste, but not the therapeutic effect.
Patient experiences vary. Some people swear by the brand-name drug, while others see no difference at all. A 2023 survey showed that 78% of patients were satisfied with generic medications. The dissatisfaction often comes from the placebo effect or genuine sensitivity to inactive ingredients like dyes or fillers. For example, a patient might react to a specific dye in a generic pill but not the active drug itself. This is an allergy or intolerance to an excipient, not a failure of bioequivalence.
Another common concern is switching between different generic manufacturers. If you go to one pharmacy and get Generic Brand A, and then go to another and get Generic Brand B, you might notice a slight difference. Both are bioequivalent to the brand-name drug, but they are not necessarily bioequivalent to each other. This is known as the "generic-to-generic" switch issue. While rare, it can happen. If you notice changes in how you feel after a switch, talk to your pharmacist about keeping a consistent manufacturer.
What This Means for Your Health
Ultimately, bioequivalence is a tool designed to protect you. It ensures that when you save money on your prescription, you aren't sacrificing safety or effectiveness. The regulatory framework is robust, backed by decades of data and real-world evidence. For most people, switching to a generic is a safe and smart financial decision.
However, you are the expert on your own body. If you have a history of sensitive reactions, or if you are taking a narrow therapeutic index drug, communicate openly with your healthcare provider. Ask questions. If a generic doesn't seem to work as well, don't suffer in silence. Your doctor can write "dispense as written" for the brand-name drug if medically necessary, though insurance might not cover the extra cost.
The goal of bioequivalence testing is to provide confidence. It bridges the gap between expensive innovation and accessible care. By understanding what the term really means, you can make informed decisions about your medication without fear or confusion. You know now that the different-looking pill in your hand has been tested to ensure it delivers the medicine your body needs, just like the original.
Are generic drugs exactly the same as brand-name drugs?
Generic drugs contain the same active ingredients, strength, and dosage form as brand-name drugs, but they may differ in inactive ingredients like colors, flavors, and preservatives. They must be bioequivalent, meaning they work the same way in the body, but they do not have to look identical.
What is the 80-125% rule in bioequivalence?
The FDA requires that the bioavailability of a generic drug falls within 80% to 125% of the brand-name drug's bioavailability. This range is statistically derived to ensure that any difference in drug exposure is not clinically significant for most medications.
Can I switch between different generic manufacturers?
Generally, yes, as both generics are bioequivalent to the brand-name drug. However, for narrow therapeutic index drugs, it is often recommended to stay with the same manufacturer to ensure consistent blood levels of the medication.
Why do some generics cost less than others?
Prices vary based on the number of manufacturers producing the drug, the cost of production, and market competition. More competition usually drives the price down, making the medication more affordable for patients.
Do I need a new prescription to get a generic?
No, in most cases, pharmacists can automatically substitute a generic for a brand-name drug without a new prescription, unless the doctor has specifically written "dispense as written" on the prescription.
Next Steps for Patients
If you have concerns about your medication, here are practical steps you can take. First, ask your pharmacist about the specific generic manufacturer they are dispensing. They can often tell you if there are multiple options available. Second, if you are on a sensitive medication like levothyroxine or an anti-seizure drug, ask your doctor if you should stick to one brand or manufacturer. Third, keep a log of how you feel after any medication change. This information is valuable for your doctor if you need to justify a switch back to a brand-name drug.
Remember, the healthcare system relies on bioequivalence to keep costs manageable. While no two pills are ever exactly identical down to the molecule, the standards in place ensure that the difference is negligible for the vast majority of patients. By understanding the science, you empower yourself to manage your health confidently, whether you are taking the original brand or a cost-effective generic alternative.







12 comments
Jefferson Moratin
The concept of bioequivalence represents a fascinating intersection between economic necessity and medical ethics. We must consider the broader implications of standardizing treatment across different socioeconomic groups. It is reassuring to see that rigorous testing protocols maintain the integrity of patient care. The distinction between active and inactive ingredients is often overlooked by the general public. Understanding these nuances empowers individuals to make informed choices about their health. Trust in the regulatory framework is essential for the stability of the healthcare system. We should appreciate the scientific rigor that underpins these approval processes. The balance between innovation and accessibility is delicate yet vital. It is important to remain vigilant about the quality of medication we consume.
Mihir Patel
My heart races when I see a diffrent pill color!!
Kevin Y.
Pharmaceutical standards require a high degree of consistency to ensure public safety. It is indeed a pleasure to read such a well-structured explanation of these protocols. We should all strive to understand the science behind our medications. The dedication to clarity is truly commendable in this field. It fosters a sense of community and shared knowledge among us. I look forward to reading more content on similar topics. We should continue to discuss these important health topics.
Raphael Schwartz
US drugs are best and no one can say diffrent.
Grace Kusta Nasralla
It feels like they dont care about our feelings when they change the pills.
Stephen Alabi
I must point out that your assumption regarding patient feelings is somewhat flawed. The regulatory bodies prioritize data over subjective emotional responses. There is a distinct difference between perceived efficacy and actual pharmacokinetic results. We should rely on empirical evidence rather than anecdotal experiences. The scientific method demands objectivity in these matters. It is crucial to maintain a rational perspective when discussing medical standards.
Agbogla Bischof
The, study, indicates, that, the, variance, is, within, acceptable, limits, and, we, should, trust, the, process.
Caroline Bonner
We, need, to, trust, the, science, behind, the, meds. It, helps, so, much, with, my, worries. I, was, worried, before, I, learned, this. Now, I, know, the, truth, about, meds. The, science, is, clear, and, simple. We, should, trust, the, FDA, completely. They, check, everything, very, carefully. It, makes, me, feel, safe, inside. My, mom, takes, generics, every, day. She, is, doing, fine, with, them. We, save, money, too, on, bills. That, is, important, for, families. Healthcare, costs, are, very, high, now. Everyone, needs, help, with, prices. We, should, share, this, info, with, others. Keep, sharing, info, with, us, all. It, is, good, for, the, community.
peter vencken
i thnk its ok to switch if u save cash.
Chris Crosson
That is a valid point regarding the financial aspect of medication. Saving money is crucial for maintaining long-term treatment adherence. We should not overlook the economic benefits of generic substitution. Your perspective adds value to this discussion.
Kevin Siewe
Patients should feel confident when they switch to a generic option. It is good to be informed before taking any medication. Please remember to talk to your doctor if you have concerns.
Chris Farley
Generic is trash and you should stick to brand.